
ADHERE: the largest CIDP trial ever conducted1,2
Discover how efficacy and safety were demonstrated for VYVGART Hytrulo in adults with CIDP.
ADHERE was a 2-stage study, with an initial treatment period (open label, Stage A) to identify responders who then proceeded to the randomized withdrawal period (placebo controlled, Stage B).1
Initial Treatment Period (Open Label, Stage A)
The majority of patients responded to VYVGART Hytrulo based on an improvement in functional ability or strength1

69% of patients improved before entering the randomized withdrawal period (n=221/322).1
The initial treatment period (up to 12 weeks) identified VYVGART Hytrulo responders who had improvement in functional ability (defined as an aINCAT improvement of ≥1 point or an I-RODS improvement of ≥4 points) or strength (defined as mean grip strength improvement of ≥8 kPa) at 2 consecutive visits.1
Randomized Withdrawal Period (Placebo Controlled, Stage B)
Significant reduction in the risk of clinical deterioration (relapse) with VYVGART Hytrulo1
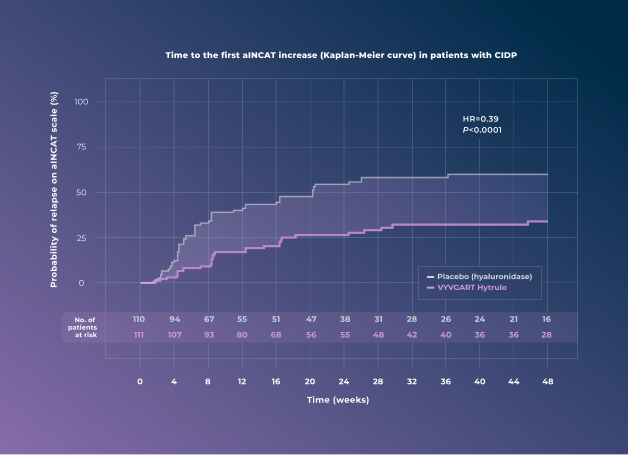
61% reduction in the risk of relapse vs placebo (HR=0.39 [95% CI: 0.25-0.61; P<0.0001])1
The primary endpoint for the randomized withdrawal period was the time to first clinical deterioration (relapse), defined as a 1-point increase in aINCAT score at 2 consecutive visits or a ≥1-point increase in aINCAT score at 1 visit from baseline at randomization.1
There were 221 patients at the beginning of the randomized withdrawal period (111 patients treated with VYVGART Hytrulo and 110 patients receiving placebo).1
VYVGART Hytrulo has a demonstrated safety profile
The overall safety profile observed in patients with CIDP treated with VYVGART Hytrulo was consistent with the known safety profile of VYVGART Hytrulo and of efgartigimod alfa-fcab administered intravenously.1
In ADHERE Stage B, infections occurred in 32% of patients treated with VYVGART Hytrulo and 34% of placebo-treated patients. The common infections were COVID-19 (17% VYVGART Hytrulo vs 13% placebo), nasopharyngitis (5% VYVGART Hytrulo vs 3% placebo), URTI (2% VYVGART Hytrulo vs 10% placebo), and pneumonia (1% VYVGART Hytrulo vs 4% placebo).2,3
In ADHERE Stage B, injection site reactions occurred in 15% of patients treated with VYVGART Hytrulo and 6% of placebo-treated patients. Injection site reactions were bruising (5% VYVGART Hytrulo vs 1% placebo) and erythema (5% VYVGART Hytrulo and 0% placebo).1-3
Please see Important Safety Information below for VYVGART Hytrulo.
Ready to learn more about the ADHERE trial? Visit https://vyvgarthcp.com/vyvgarthytrulo-cidp/clinical-data
aINCAT=adjusted Inflammatory Neuropathy Cause and Treatment; CIDP=chronic inflammatory demyelinating polyneuropathy; HR=hazard ratio; I-RODS=Inflammatory Rasch‐built Overall Disability Scale; kPa=kilopascals; URTI=upper respiratory tract infection.
IMPORTANT SAFETY INFORMATION AND INDICATION
CONTRAINDICATIONS
VYVGART HYTRULO is contraindicated in patients with serious hypersensitivity to efgartigimod alfa products, to hyaluronidase, or to any of the excipients of VYVGART HYTRULO. Reactions have included anaphylaxis and hypotension leading to syncope.
WARNINGS AND PRECAUTIONS
Infections
VYVGART HYTRULO may increase the risk of infection. The most common infections observed in Study 1 in patients with gMG were urinary tract infection (10% of efgartigimod alfa-fcab-treated patients vs 5% of placebo-treated patients) and respiratory tract infections (33% of efgartigimod alfa-fcab-treated patients vs 29% of placebo-treated patients). Patients on efgartigimod alfa-fcab vs placebo had below normal levels for white blood cell counts (12% vs 5%, respectively), lymphocyte counts (28% vs 19%, respectively), and neutrophil counts (13% vs 6%, respectively). The majority of infections and hematologic abnormalities were mild to moderate in severity. Delay VYVGART HYTRULO administration in patients with an active infection until the infection has resolved; monitor for clinical signs and symptoms of infections. If serious infection occurs, administer appropriate treatment and consider withholding VYVGART HYTRULO until the infection has resolved.
Immunization
Evaluate the need to administer age-appropriate vaccines according to immunization guidelines before initiation of a new treatment cycle with VYVGART HYTRULO. The safety of immunization with live vaccines and the immune response to vaccination during treatment with VYVGART HYTRULO are unknown. Because VYVGART HYTRULO causes a reduction in immunoglobulin G (IgG) levels, vaccination with live vaccines is not recommended during treatment with VYVGART HYTRULO.
Hypersensitivity Reactions
In clinical trials, hypersensitivity reactions, including rash, angioedema, and dyspnea were observed in patients treated with VYVGART HYTRULO or intravenous efgartigimod alfa-fcab. Urticaria was also observed in patients treated with VYVGART HYTRULO. Hypersensitivity reactions were mild or moderate, occurred within 1 hour to 3 weeks of administration. Anaphylaxis and hypotension leading to syncope have been reported in postmarketing experience with intravenous efgartigimod alfa-fcab. Anaphylaxis and hypotension occurred during or within an hour of administration and led to infusion discontinuation and in some cases to permanent treatment discontinuation. Monitor for clinical signs and symptoms of hypersensitivity reactions for at least 30 minutes after administration. If a hypersensitivity reaction occurs, the healthcare professional should institute appropriate measures if needed or the patient should seek medical attention.
Infusion/Injection-Related Reactions
Infusion-related reactions have been reported with intravenous efgartigimod alfa-fcab in postmarketing experience. The most frequent symptoms and signs were hypertension, chills, shivering, and thoracic, abdominal, and back pain. Infusion-related reactions occurred during or within an hour of administration and led to infusion discontinuation. If a severe infusion/injection-related reaction occurs, initiate appropriate therapy. Consider the risks and benefits of readministering VYVGART HYTRULO following a severe infusion/injection-related reaction. If a mild to moderate infusion/injection-related reaction occurs, patients may be rechallenged with close clinical observation, slower infusion/injection rates, and pre-medications.
ADVERSE REACTIONS
Patients with gMG: In Study 1, the most common (≥10%) adverse reactions in efgartigimod alfa-fcab-treated patients were respiratory tract infection, headache, and urinary tract infection. In Study 2, the most common (≥10%) adverse reactions in VYVGART HYTRULO-treated patients were injection site reactions and headache. Injection site reactions occurred in 38% of VYVGART HYTRULO-treated patients, including injection site rash, erythema, pruritus, bruising, pain, and urticaria. In Study 2 and its open-label extension in patients with gMG, all injection site reactions were mild to moderate in severity and did not lead to treatment discontinuation. The majority occurred within 24 hours after administration and resolved spontaneously. Most injection site reactions occurred during the first treatment cycle, and the incidence decreased with each subsequent cycle.
Patients with CIDP: In Study 3 stage B, the overall safety profile observed in patients with CIDP treated with VYVGART HYTRULO was consistent with the known safety profile of VYVGART HYTRULO and of efgartigimod alfa-fcab administered intravenously. In Study 3, injection site reactions occurred in 15% of patients treated with VYVGART HYTRULO compared to 6% of patients who received placebo. The most common of these injection site reactions were injection site bruising and injection site erythema. All injection site reactions were mild to moderate in severity. Most injection site reactions occurred during the first 3 months of treatment.
USE IN SPECIFIC POPULATIONS
Pregnancy
As VYVGART HYTRULO is expected to reduce maternal IgG antibody levels, reduction in passive protection to the newborn is anticipated. Risk and benefits should be considered prior to administering live vaccines to infants exposed to VYVGART HYTRULO in utero.
Lactation
There is no information regarding the presence of efgartigimod alfa or hyaluronidase, from administration of VYVGART HYTRULO, in human milk, the effects on the breastfed infant, or the effects on milk production. Maternal IgG is known to be present in human milk. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for VYVGART HYTRULO and any potential adverse effects on the breastfed infant from VYVGART HYTRULO or from the underlying maternal condition.
INDICATION
VYVGART HYTRULO® (efgartigimod alfa and hyaluronidase-qvfc) is indicated for the treatment of adult patients with generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) antibody positive.
VYVGART HYTRULO® (efgartigimod alfa and hyaluronidase-qvfc) is indicated for the treatment of adult patients with chronic inflammatory demyelinating polyneuropathy (CIDP).
Please see the full Prescribing Information.
You may report side effects to the US Food and Drug Administration by visiting http://www.fda.gov/medwatch or calling 1-800-FDA-1088. You may also report side effects to argenx US, Inc, at 1-833-argx411 (1-833-274-9411).
Dosage Forms and Strengths: VYVGART Hytrulo is available as a single-dose subcutaneous injection containing: 200 mg/mL of efgartigimod alfa and 2,000 U/mL of hyaluronidase per prefilled syringe, or 180 mg/mL of efgartigimod alfa and 2,000 U/mL of hyaluronidase per vial.
References: 1. VYVGART Hytrulo. Prescribing information. argenx US Inc; 2025. 2. Allen JA et al. Lancet Neurol. 2024;23(10):1013-1024. doi:10.1016/S1474-4422(24)00309-0 3. Allen JA et al. Supplementary appendix. Lancet Neurol. 2024;23(10):1-43. doi:10.1016/S1474-4422(24)00309-0
For US audiences only.
VYVGART Hytrulo is a registered trademark of argenx.
© 2026 argenx US-VYV_HYT-25-00528 V1 01/2026.
All Rights Reserved.
-
 Sherri Hernandez1wI was unaware there were treatments for CIPD. This progressive disease is debilitating. Vygart seems promising.
Sherri Hernandez1wI was unaware there were treatments for CIPD. This progressive disease is debilitating. Vygart seems promising. -
 Temeki Hill1wno i was not aware but glad i am now, this is a very good treatment option for this disease and like pretty much all biologics (similar meds) the side/adverse effects are all the same but showed really no clinical difference between the active group versus placebo group and the local effects are minimal compared to the disease itself and its progression
Temeki Hill1wno i was not aware but glad i am now, this is a very good treatment option for this disease and like pretty much all biologics (similar meds) the side/adverse effects are all the same but showed really no clinical difference between the active group versus placebo group and the local effects are minimal compared to the disease itself and its progression -
 Mike Shirk2wi was not aware of the trial data of Vyvgart. The efficacy in improving CIDP symptoms is impressive with 7/10 patient showing improvement and also 61 percent reduction in relapse. i expect to increase my prescribing
Mike Shirk2wi was not aware of the trial data of Vyvgart. The efficacy in improving CIDP symptoms is impressive with 7/10 patient showing improvement and also 61 percent reduction in relapse. i expect to increase my prescribing -
 Anjaneyulu Alapati1moconfident to prescribe. robust results. effective and safe compared to placebo. my prescriptions will go up
Anjaneyulu Alapati1moconfident to prescribe. robust results. effective and safe compared to placebo. my prescriptions will go up -
 Jonathan Lee1mothis trial demonstrates convincing evidence of an effective and convenient alternative to IVIG or steroids for CIDP. Its the largest randomized controlled study for CIDP.
Jonathan Lee1mothis trial demonstrates convincing evidence of an effective and convenient alternative to IVIG or steroids for CIDP. Its the largest randomized controlled study for CIDP. -
 Ho Sheng Lin1moPromising data and look forward to hearing more about it in the near future.
Ho Sheng Lin1moPromising data and look forward to hearing more about it in the near future. -
 Paola Sandroni1moI am familiar with the data and although we were initially enthusiastic to have another options and a convenient options particularly with the Vyvgart hytrulo, the response rate has not been as good as with standard of care with IVIG and similar. Indeed we had couple of patient that reverted back.
Paola Sandroni1moI am familiar with the data and although we were initially enthusiastic to have another options and a convenient options particularly with the Vyvgart hytrulo, the response rate has not been as good as with standard of care with IVIG and similar. Indeed we had couple of patient that reverted back. -
 MICHAEL SHIRK1moi was not aware of this medication prior but the Vyvgart data shows efficacy in CIDP patients and will consider its use in patients
MICHAEL SHIRK1moi was not aware of this medication prior but the Vyvgart data shows efficacy in CIDP patients and will consider its use in patients -
 Jorge Osorio1moI am not familiar with this medication and trials but It appears a promising treatment for CIDP.
Jorge Osorio1moI am not familiar with this medication and trials but It appears a promising treatment for CIDP. -
 Terry Cheung1moI've seen it before and looked into this specific trial. Vyvgart does show a promising data for CIDP patients and that is definitely a breakthrough in CIDP treatments.
Terry Cheung1moI've seen it before and looked into this specific trial. Vyvgart does show a promising data for CIDP patients and that is definitely a breakthrough in CIDP treatments. -
 JOEL WASHINSKY1moAm familiar and it looks to be quite efficacoius.
JOEL WASHINSKY1moAm familiar and it looks to be quite efficacoius. -
 SACHINKUMAR PANCHAL1moRead about this trial and need FDA approval and need real world data before using it
SACHINKUMAR PANCHAL1moRead about this trial and need FDA approval and need real world data before using it -
 Rahat Sheikh1moI was unfamiliar of this trial but I think it is quite promising AND WILL READILY USE IT
Rahat Sheikh1moI was unfamiliar of this trial but I think it is quite promising AND WILL READILY USE IT -
 Crystal Worsena1moaware of this trial and looking forward to the indication for use and CIDP
Crystal Worsena1moaware of this trial and looking forward to the indication for use and CIDP -
 Brandon Lucke-Wold1moUnfamiliar but favorable results for those that would be cleared from increased infection standpoint.
Brandon Lucke-Wold1moUnfamiliar but favorable results for those that would be cleared from increased infection standpoint. -
 Millie Burke1moI was unfamiliar with this trial. These look extremely viable for patients especially regarding relapse reduction and improving functionality in patients.
Millie Burke1moI was unfamiliar with this trial. These look extremely viable for patients especially regarding relapse reduction and improving functionality in patients. -
 Liang Lu2moI am familiar with the ADHERE trial results, which demonstrated that Vyvgart Hytrulo significantly reduced the risk of relapse in CIDP vs placebo, meeting its primary endpoint. Patients also demonstrated meaningful improvements in disability and strength, with a safety profile consistent with prior experience of Vyvgart.
Liang Lu2moI am familiar with the ADHERE trial results, which demonstrated that Vyvgart Hytrulo significantly reduced the risk of relapse in CIDP vs placebo, meeting its primary endpoint. Patients also demonstrated meaningful improvements in disability and strength, with a safety profile consistent with prior experience of Vyvgart. -
 ANDREW ROGOVE2moI had not been familiar with the results of this trial. It’s pretty impressive at how well Vyvgart seems to work for CIDP and how the effective sustained overtime with a randomized withdrawal. also, the safety ad adverse effect data presented seem to be decent, especially when compared to placebo
ANDREW ROGOVE2moI had not been familiar with the results of this trial. It’s pretty impressive at how well Vyvgart seems to work for CIDP and how the effective sustained overtime with a randomized withdrawal. also, the safety ad adverse effect data presented seem to be decent, especially when compared to placebo -
 Victoria Bradley2moAre you familiar with the data from the ADHERE trial in CIDP? Share your thoughts in the comments!
Victoria Bradley2moAre you familiar with the data from the ADHERE trial in CIDP? Share your thoughts in the comments!

.png)



