
Circulating GPIHBP1 levels and microvascular complications in patients with type 2 diabetes: A cross-sectional study
Source : https://www.lipidjournal.com/article/S1933-2874(22)00021-6/pdf
Dyslipidemia is a major risk factor for coronary heart disease (CHD) in patients with type 2 diabetes mellitus (T2DM),1,2 whose risk of CHD is two-to three-fold higher than in individuals...
Highlights: "Serum GPIHBP1 levels were higher in patients with type 2 diabetes mellitus (T2DM). Serum GPIHBP1 levels were higher in T2DM patients with microvascular complications. Serum GPIHBP1 levels independently associated with T2DM microvascular complications".
Highlights: "L. plantarum ECGC 13110402 supports the reduction of multiple cholesterol biomarkers. L. plantarum ECGC 13110402 has high bile salt hydrolase activity. L. plantarum ECGC 13110402 is a well-tolerated and safe probiotic strain. L. plantarum ECGC 13110402 improves the lipid profiles of dyslipidaemic patients".
In conclusion, coronary CTA has provided welcome insights into the role of Lp(a) in plaque progression. In concert with imaging evidence, the clinical evidence on whether substantially lowering Lp(a) will reduce cardiovascular events is expected in 2025 from Lp(a) HORIZON. A positive trial is likely to lead to additional trials and new drugs...
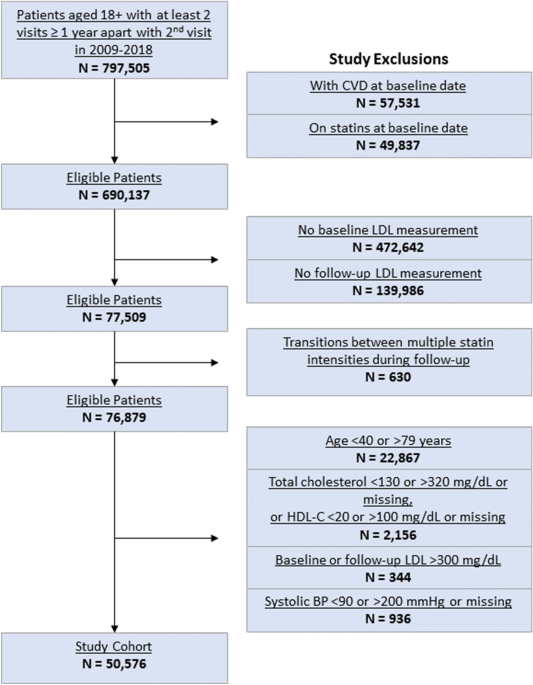
Personalizing cholesterol treatment recommendations for primary cardiovascular disease prevention - Scientific Reports
Source : https://www.nature.com/articles/s41598-021-03796-6
Statin therapy is the cornerstone of preventing atherosclerotic cardiovascular disease (ASCVD), primarily by reducing low density lipoprotein cholesterol (LDL-C) levels. Optimal statin therapy decisions rely on shared decision making and...
Recommendations included low- or moderate-intensity statins for maximum LDL-C lowering in nearly half the test set, which is discordant with their expected guideline-based efficacy. A data-driven personalized statin recommendation approach may inform shared decision making in areas of uncertainty, and highlight unexpected efficacy-effectiveness...

FDA Clears Novartis Inclisiran to Lower Cholesterol With Only Two Doses a Year
January 4, 2022 - The U.S. Food and Drug Administration (FDA) has cleared Novartis' inclisiran (Leqvio), the first small interfering RNA (siRNA) therapy to lower low-density lipoprotein cholesterol (LDL-C) with...
“Leqvio is a revolutionary approach to lower LDL-C, and creates new possibilities for how healthcare systems can impact cardiovascular disease, a defining public health challenge of our time,” said Vas Narasimhan, Novartis CEO. “We now have the opportunity, working together with partners, to provide this first-ever approved...






