Efficacy and safety of GLP-1 receptor agonists versus SGLT-2 inhibitors in overweight/obese patients with or without diabetes mellitus: a systematic review and network meta-analysis
Source : https://bmjopen.bmj.com/content/13/3/e061807
Objective To compare the efficacy and safety between and within glucagon-like peptide-1 receptor agonists (GLP-1RAs) and sodium-glucose cotransporter 2 inhibitors (SGLT-2is) in overweight or obese adults with or without diabetes...
Conclusions: Semaglutide 2.4 mg showed the greatest effects on losing body weight, controlling glycaemic level and reducing blood pressure while it was associated with high risk of adverse events.

Effect of once-weekly subcutaneous semaglutide 2.4 mg on weight- and health-related quality of life in an East Asian population: Patient-reported outcomes from the STEP 6 trial - PubMed
Source : https://pubmed.ncbi.nlm.nih.gov/36905345/
We assessed the effect of semaglutide 2.4 and 1.7 mg versus placebo on weight-related quality of life (WRQOL) and health-related quality of life (HRQOL) in the STEP 6 trial. Adults...
Conclusion: Overweight and obesity can have a negative impact on WRQOL and HRQOL. Our results demonstrate that in people from East Asia with overweight or obesity, treatment with once-weekly subcutaneous semaglutide 2.4 mg, in addition to a reduced caloric diet and increased physical activity, improved aspects of WRQOL and HRQOL,...
-
 Endocrinologist Nation2yrKey Points • Source: Clinical Obesity • Conclusions/Relevance: “Treatment with semaglutide 2.4 mg improved aspects of WRQOL [work-related quality of life] and HRQOL [health-related quality of life] in people from East Asia with overweight/obesity.” • In Show More
Endocrinologist Nation2yrKey Points • Source: Clinical Obesity • Conclusions/Relevance: “Treatment with semaglutide 2.4 mg improved aspects of WRQOL [work-related quality of life] and HRQOL [health-related quality of life] in people from East Asia with overweight/obesity.” • In Show More
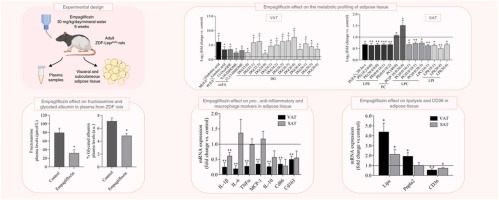
The lipidomic and inflammatory profiles of visceral and subcutaneous adipose tissues are distinctly regulated by the SGLT2 inhibitor empagliflozin in Zucker diabetic fatty rats
Source : https://www.sciencedirect.com/science/article/pii/S0753332223003232?via=ihub
Empagliflozin distinctly modulates the lipidome of rat white adipose tissue depots. * This drug induces a strong anti-inflammatory effect in rat visceral fat. * Empagliflozin reduces M1 and M2 macrophage...
Conclusions/Relevance: Our data highlighted differences in the VAT and SAT lipidomes, inflammatory profiles and lipolytic function, which suggest a distinct metabolism of these two white adipose tissue depots after the empagliflozin treatment.

GLP-1/GIP analogues: potential impact in the landscape of obesity pharmacotherapy - PubMed
Source : https://pubmed.ncbi.nlm.nih.gov/36927378/
The .gov means it's official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you're on a federal government site. The site is secure....
Expert Opinion: GLP-1R mimetics are often discontinued within 24-months, owing to gastrointestinal side-effects, meaning long-term application of these agents in obesity is questioned. Combined GIPR/GLP-1R agonism appears to induce fewer side-effects, indicating GLP-1R/GIPR co-agonists may be more suitable for enduring obesity management....

Effect of Various Dosing Schedules on the Pharmacokinetics of Oral Semaglutide: A Randomised Trial in Healthy Subjects - Clinical Pharmacokinetics
Source : https://link.springer.com/article/10.1007/s40262-023-01223-9
Background Prescribing information instructs taking oral semaglutide (a glucagon-like peptide-1 analogue) in the fasting state, followed by a post-dose fasting period of ≥ 30 min. This trial compared the recommended dosing...
Conclusion: This trial supports dosing oral semaglutide in accordance with prescribing information, which requires dosing in the fasting state.





